Clinical Resource: A Series of Evaluations Assessing the Efficacy of ActivHeal® Hydrocolloid
Margaret Caddy
Registered Nurse, Wallingford Community Hospital,
Oxfordshire PCT, Wallingford
Clinical Resource
A Series of Evaluations Assessing the Efficacy of ActivHeal® Hydrocolloid
Margaret Caddy
Registered Nurse, Wallingford Community Hospital, Oxfordshire PCT, Wallingford
INTRODUCTION
There are a wide variety of wound care dressings available in the market place. All of these dressings vary in relation to evidence and performance characteristics. We underwent a case study to assess the efficacy of the ActivHeal® Hydrocolloid dressing. A specific patient evaluation has been remarked upon to substantiate the product performance and will be described as Patient X.
Hydrocolloid dressings were developed from stoma products and most consist of a base made from pectin or gelatin, sodium carboxymethyl cellulose and elastomers. The base is attached to a polyurethane film or backing. ActivHeal® Hydrocolloid contains a blend of synthetic polymers and hydrophilic powders which provides a self adhesive absorbent material for wound application, not derived from animal property. The dressing when in situ is designed to encourage autolysis and absorbs fluid to form a cohesive gel.
METHOD
ActivHeal® Hydrocolloid was evaluated on seven patients over a four to eight week period. At each dressing change a wound assessment was completed detailing: tissue type at the wound bed, surrounding skin, exudate levels, wound dimensions and wound pain. The assessing clinician commented on dressing adherence, ease of application, conformability, absorbency, patient comfort, dressing residue left in the wound and slippage. The assessment was completed once a week for the four to eight week period. Photographs of the wound state were taken at each dressing change to substantiate dressing performance.
RESULTS
The results gained from the dressing performance were collated and analysed. Three of the wounds healed in four weeks from commencing therapy and two wounds showed a reduction in wound dimension following dressing intervention. The assessing clinician highlighted that all of the applications were easy to apply, conformed to the affected area, absorbed exudate, provided good standards of patient comfort, did not adhere to the wound bed following removal and stayed in situ for the indicated period, graph one demonstrates this.
Graph 1
Graph 2
Patient X was referred for wound care intervention and presented with a below knee surgical wound following amputation to the right leg. Patient X also presented with skin breakdown to the left leg and queried possible underlying venous abnormality. Both wounds were managed using ActivHeal® Hydrocolloid. The post surgical wound to the right leg reduced in size from 8cms and fully healed within four weeks, photograph 1a and 1b reflects this.
The wound to the left leg reduced in dimension by 2.5cm and became less sloughy following granulation promotion, the wound did not heal fully due to queried underlying co-morbidities and graph 2 outlines this result. Further to this, photographs 2a and 2b reflect progress.
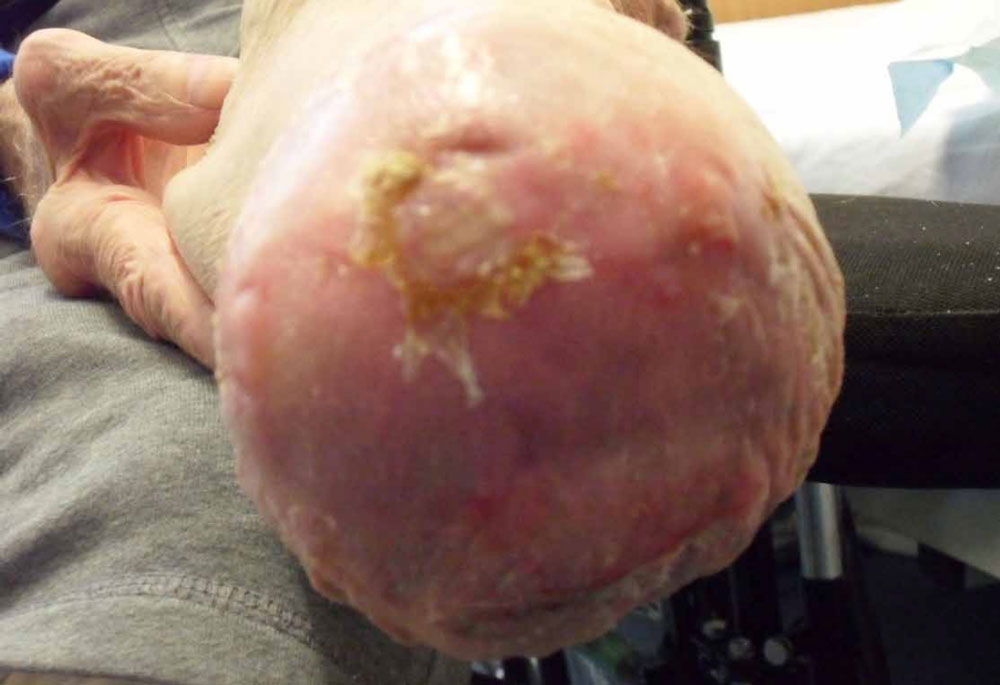
Photograph 1a
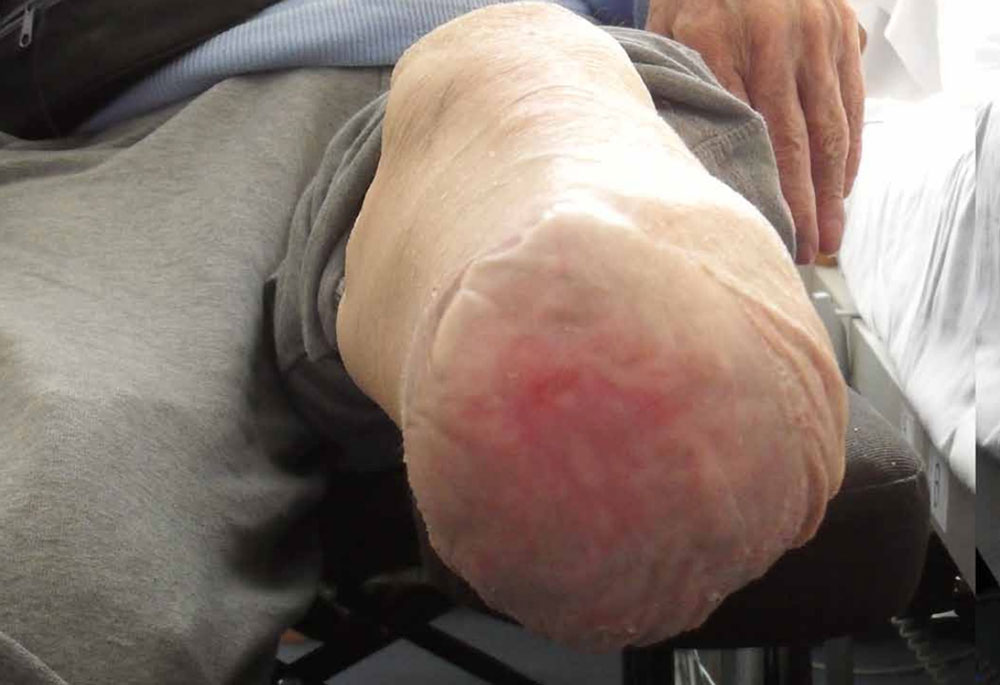
Photograph 1b
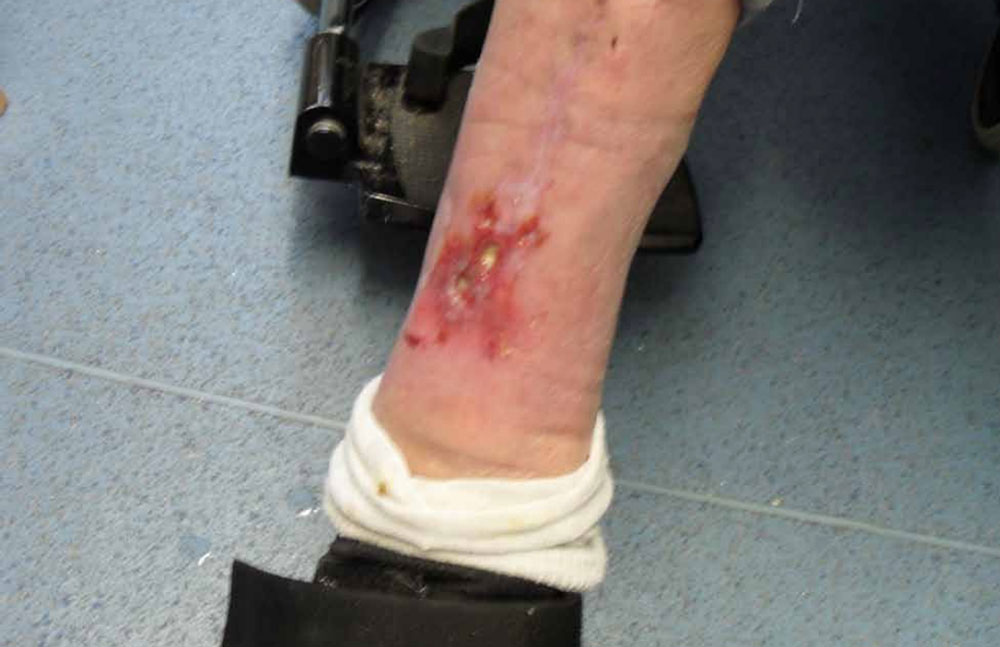
Photograph 2a
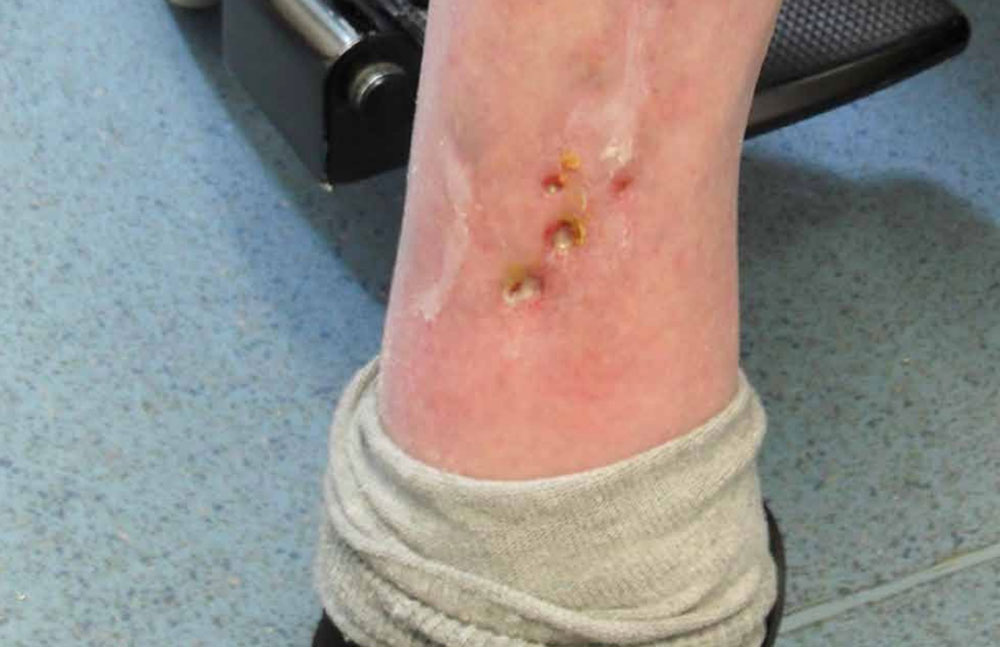
Photograph 2b
DISCUSSION
There is much controversy in relation to driving cost down in the NHS whilst not compromising patient care. ActivHeal® Hydrocolloid offers significant savings over leading branded hydrocolloids, yet delivers positive clinical outcomes. The ActivHeal® range is being used in both the acute and primary care settings and is widely acknowledged as being a first line choice of dressings post wound care assessment. Justification for change to the ActivHeal® range should be based around positive clinical outcomes and cost effective solutions.
CONCLUSION
ActivHeal® Hydrocolloid is a good choice for trusts wanting to reduce cost and maintain effective patient centered care.
CONTACT US FOR MORE INFORMATION
Discover ActivHeal®
Social Media
Our Product Range
AMS Group
ActivHeal®, its logo and the Advanced Medical Solutions logos are registered trademarks of Advanced Medical Solutions Ltd.
Copyright © Advanced Medical Solutions Limited | Design by Lumisi Ltd